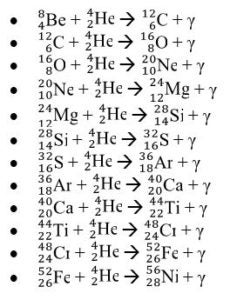Three lessons have been designed in order to enhance student learning in the classroom. The first lesson focuses on engaging the student and using what the student knows about his personal identity in order to gain insight into the identity of atoms. The second lesson uses nuclear notation to write nuclear equations that tell the story of stellar nucleation. The third lesson utilizes a graphical approach to show students that nuclear composition gradually changes as elements become heavier, indicating that other processes are necessary to produce heavier elements. Supplemental materials and ideas are provided at the conclusion of the third lesson.
Lesson: Discover Your Atomic Identity
Purpose: The purpose of this lesson is for students to understand how the subatomic particles influence the identity of atoms. This lesson will require two days of classroom time. Students will examine their own identities first. Then they will learn about the identity of atoms using prompts, lists, and an expert jigsaw activity.
Instructional Objectives: At the end of this lesson, students will be able to describe the evidence for the existence of electrons, protons, and neutrons in order to describe the properties of these subatomic particles.
Materials and Equipment: Chart paper, textbooks, computer with internet access, and markers.
Engage: Students will be prompted to create 3 lists with each list having 3 activities or actions that they personally perform. The three prompts are each related to the three subatomic particles and their function. The students may remember the subatomic particles from middle school, but may not necessarily understand their function. Therefore, this activity focuses heavily on using their reflections as gateways to understanding the functions of the subatomic particles. Begin the lesson by asking the students to Think-Pair-Share-Write after each prompt below is presented.
- Name 3 activities that you do that make you absolutely unique and incomparable to any other human being. Think about activities that if you were to stop doing them today, you would be a completely different human being. This prompt is designed for students see in themselves what makes them stand out and unique. Answers may vary incredibly depending on what each student values. For example, a student might take pride in wearing the latest pair of neon green shoes, while another student might mention a specific skill that they believe they have mastered. At the end of the lesson, students should be able to understand that the uniqueness of each atom is characterized by the protons.
- Name 3 activities that you do by yourself to either calm down or relax in order to keep your sanity. This prompt is designed to help students understand the function of neutrons. Again, answers may vary depending on what a student values. For example, some students might say that eating cake or ice cream is a ritual that they do to maintain stability in their lives. Others might mention playing music, or exercising. Neutrons are responsible for stabilizing the nucleus, or stabilizing the identity of the atom. At the end of the lesson, students should be able to understand that neutrons are essential in stabilizing the nucleus just as the activities listed for this specific prompt are to stabilize the human soul and to secure or fortify the identity.
- Name 3 actions that you would perform in order to maintain good relationships with people. This prompt is designed to help students understand the function of the electrons. Answers may vary from buying gifts for their family and friends, giving hugs, acts of service, etc. They need to grasp that electrons are responsible for how atoms of different identities interact with one another. Thus, the students are expected to find similarities between human interactions and interactions between atoms. Students ought to realize that unselfish actions are necessary to maintain good relationships just as electrons are necessary for atoms to form stable relationships in the form of bonds.
Subatomic Particles Jigsaw and Gallery Walk: Students will engage in a subatomic particles Jigsaw. Using the textbook and online resources, students will get into 8 groups. For large classes, each topic for the jigsaw will be assigned twice. Each group conduct research on a specific subatomic particle. Afterwards, students will go back to their home groups teach the information to their peers. The groups are named as follows:
- Protons
- Neutrons
- Electrons
- Mass Number/Isotopes
Rules to the Jigsaw and Gallery Walk
- Must draw a poster that helps explain your piece of the jigsaw. It cannot just be a list of facts.
- Select a group member who will teach the classmates during the gallery walk.
- Each piece of the jigsaw requires a non-linguistic representation in the form of a bodily movement. This movement needs to be taught to classmates as well.
- If you are not selected to teach during the gallery walk, you are to take notes from the other groups.
- At the end of the gallery walk, you will return to your home group and make sure the group member that you selected to be the teacher obtains all the notes that you took.
Students should be assessed to determine how well they educated one another with your guidance. The subsequent lesson(s) should include quick lab experiments that verify the existence of the subatomic particles and instruction in regards to calculating the number of subatomic particles in each unique atom or isotope.
Lesson: Nuclear Notation
Purpose: The purpose of this lesson is for students to learn how scientists communicate atoms through literature on an international level. Students will learn nuclear notation and will be able to identify individual isotopes based on their mass and atomic numbers. This lesson should be taught after students have learned how to calculate the number of subatomic particles in an atom given the atomic number and/or the mass number.
Materials & Equipment: PowerPoint slides, smart board, projector, colored chalk or white board pens, printed diagrams, isotope puzzle cubes made using origami.[1]
Do Now Activity: Isotope Puzzle Cubes
- Have students work in pairs. Give each pair of students an isotope puzzle cube, in which either the atomic number or the atomic symbol is missing from its symbol notation.
- Give the students 7 minutes to solve the puzzle. Some students may believe that some of the numbers are wrong. If there are students that finish early, hand them a second, and more challenging isotope puzzle cube with different elements.
- At the end of the activity, introduce the lesson objectives, and poll the students for the answers to the isotope puzzles.
- Ask the following questions to remind students to use the answers from the isotope cube to support their response:
- Can atoms of the same element have different masses? (i.e. oxygen-18 and oxygen-16)
- Can atoms of the same element have different atomic numbers?
- Can atoms of different elements have the same mass? (i.e. fluorine-18 and oxygen-18)
- Reveal to students that isotopes are atoms that retain the same number of protons, but have a varying number of neutrons.

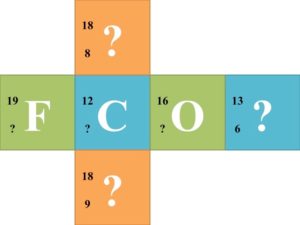
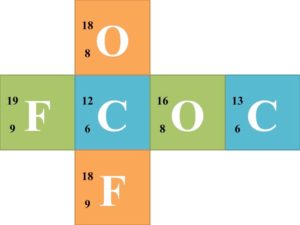
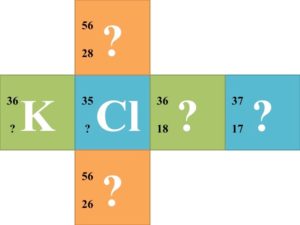
Group Activity: Divide the class into groups of six and supply each group with a cube that a nuclear symbol written on each face. Each nuclear symbol should be missing one of the three parts of the symbol, such as in the following examples: 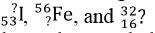 Each group of 6 should try to solve for each missing component of the nuclear symbol. Once each group finishes the 6 nuclear symbols, they should trade cubes with another group. Have the groups continue trading until every group has completed the symbols of every other group.
Each group of 6 should try to solve for each missing component of the nuclear symbol. Once each group finishes the 6 nuclear symbols, they should trade cubes with another group. Have the groups continue trading until every group has completed the symbols of every other group.
Lesson: Stellar Nucleosynthesis
Instructional Objectives: At the end of this lesson, the students will be able to define alpha process and discuss nuclides in terms of their number of subatomic particles in order to explain how elements are created naturally, i.e. nucleosynthesis, and artificially.
Engage: In order to create interest and generate curiosity, students will watch The Science of the Avengers Video[2], and journal their response to an instructional prompt as their kick-off activity, beginning at time 1:00 and ending at 1:40. The strength of this video lies in its reference to Tony Stark and his ability to synthesize a new element when he realizes that the arc reactor biologically built into his chest used to power his suit is also poisoning his body. The goal is to engage students to the idea of why scientists desire to create and discover new elements. In addition, they can even see that Hollywood writers need to consult chemists in order to write scientifically valid storylines for superheroes.
Instructional Prompt: Watch the video about the Science of the Avengers. Pay attention to the chemistry involved and answer the following questions.
- Which element was the original power source of Iron Man’s arc reactor?
- What type of equipment did Tony Stark use to create a brand new atom to power his suit?
- Why did Stark choose to create a brand new element? Think of an additional reason as to why anyone would want to manufacture elements.
Poll the classroom for answers to the questions. Pose the following essential question, derived from the inquiry statement for the unit: How are elements synthesized both naturally and synthetically in order to understand the universe in which we live in and make technological advancements?
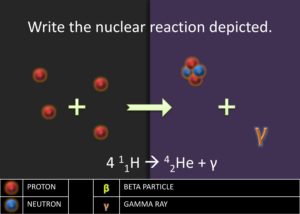
Explore: Teacher will encourage students to work together and explain a diagram of 4 protons combining in a nuclear reaction. Ask students to predict what element would form if 4 protons to fuse together. Many students will assume that a nucleus with 4 protons would have to be the beryllium-4 nuclide, 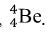 .
.
Explain: After students report out their answer, reveal the true answer that the fusion of 4 protons actually produces the helium-4 nuclide, 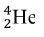 . Ask the students what could have happened to the 2 protons. Students ought to respond saying that two protons must have become neutrons. Afterward, introduce the students to gamma emission, which is the energy released as photon when a proton transforms into a neutron. Explain to students that this process is the beginning of stellar nucleation (star creation) and that the first step of stellar nucleation produces the alpha particle, which is key in the alpha process. Model for the students how to write a nuclear reaction equation using the four protons.
. Ask the students what could have happened to the 2 protons. Students ought to respond saying that two protons must have become neutrons. Afterward, introduce the students to gamma emission, which is the energy released as photon when a proton transforms into a neutron. Explain to students that this process is the beginning of stellar nucleation (star creation) and that the first step of stellar nucleation produces the alpha particle, which is key in the alpha process. Model for the students how to write a nuclear reaction equation using the four protons.
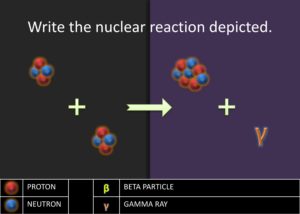
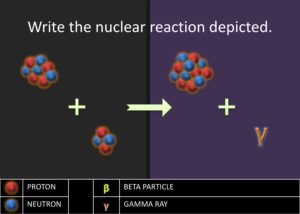
Elaborate: Show the students the next step in the stellar nucleation, using alpha process, in which two alpha particles fuse together to form the beryllium-8 nuclide, 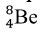 . The equation is as follows:
. The equation is as follows: 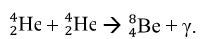 . Then ask to students to write the nuclear chemical reaction of the nuclides involved in the diagram. Have the students report out their answer and explain why they believe the nuclides they have written are what matches in the diagrams. Challenge the students to write a rule or journal the steps involved in writing the nuclear notation for nuclides produced in the alpha process. Students should have a procedure similar to the following steps:
. Then ask to students to write the nuclear chemical reaction of the nuclides involved in the diagram. Have the students report out their answer and explain why they believe the nuclides they have written are what matches in the diagrams. Challenge the students to write a rule or journal the steps involved in writing the nuclear notation for nuclides produced in the alpha process. Students should have a procedure similar to the following steps:
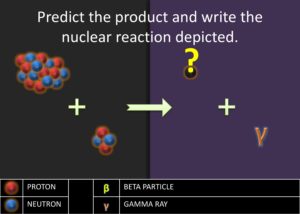
Predicting the Products of a Nuclear Reaction
- If given a diagram, count the number of protons and neutrons for each nuclide.
- Write the nuclear notation for each nuclide on the reactants side of the nuclear equation.
- Add the mass numbers of the reactants.
- Add the atomic numbers of the reactants.
- Identify the new nuclear symbol using the newly calculated atomic number.
- Write the nuclear notation for the new nuclide.
Evaluate: After showing the students the first three steps of stellar nucleation, they ought to be ready to predict the rest of the cycle using the alpha process without the use of a diagram. Pay close attention to students who may confuse the superscript and subscripts.
Nuclear Reactions and their Products in Stellar Nucleosynthesis
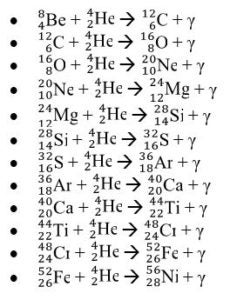
Lesson: Discovering Nuclear Stability
Purpose: The purpose of this lesson is to continue to give students practice with nuclear notation and counting protons and neutrons, while reviewing the limitations of the alpha process. Students will be led to discover that the alpha process is incapable of producing every element on the periodic table. This lesson will allow for the introduction of supernovae explosions and will bridge their learning from nuclear fusion to nuclear decay. Students will explore nuclear stability by graphing the number of neutrons vs. protons and calculating the slope of a group of elements using a graphing calculator.
Instructional Objective: Students will be able to discuss atoms in terms of their protons and neutrons in order to graph and define the relationship between the number of neutrons and protons in the nucleus as mass number increases.
Materials and Equipment: periodic tables, graphing calculators, large graph/chart paper with labeled axes (protons on x-axis, neutrons on y-axis), markers/colored pencils.
Introduction: Use the following questions as a do-now activity to or kick-off into the lesson.
- Is the atomic mass for every element always twice the number of protons?
- Can the alpha process create every element? Give an example to support your answers.
Allow the students to share their answers with the class. Student responses will vary depending on the elements that they chose as their examples. Inform the students that they will begin to explore the relationship between the number of neutrons and protons changes as more elements are added to the periodic table.
Group Activity: Divide students into groups of 3 for a total of 9 groups. Assign each group 5 elements on the periodic table. They are to plot them on a graph and calculate the slope of the line that they make. Then allow one member from each group to plot the points on a class graph. As each group adds their data points, they should see that the slope of the lines gradually increases.
Procedures:
- Obtain a list of elements from your teacher.
- Calculate the number of neutrons in each element from your list.
- Fill in the data table with the correct number of protons and neutrons for your 5 elements.
- Go to www.Desmos.com.
- Click on the [+] sign in Desmos and then click “table”.
- In the x1 column, list the atomic number of the elements from your list.
- In the y1 column, list the number of neutrons of the elements from your list.
- Click on the [+] sign in Desmos and then click “f(x) expression”.
- Type the following expression into the box: y1 ~ ax1.
- In your “f(x) expression”, you should see Statistics, Residuals, and Parameters appear if you successfully typed in the expression.
- Record the value of “a” for your list of 5 elements.
- Send a group member to the class graph to plot your group’s data points and a-value.
- Complete the analysis questions.
List of Elements for Each Group
- List 2: C, N, O F, Ne
- List 3: Si, P, S, Cl, Ar
- List 4a: Ge, As, Se, Br, Kr
- List 4b: Sc, Ti, V, Cr, Mn
- List 5a: Sn, Sb, Te, I, Xe
- List 5b: Y, Zr, Nb, Mo, Tc
- List 6a: Pb, Bi, Po, At, Rn
- List 6b: La, Ce, Pr, Nd, Pm
- List 7: Fl, Mc, Lv, Ts, Og
Analysis: As students finish entering their data into a graphing software and obtain the parameters, they will answer the following analysis questions.
- What is the value of “a” from your parameter list? Please provide 2 decimal places. Student answers will vary depending on the elements assigned to them.
- What mathematical term would you ascribe to “a” in terms of the line that you graphed? Students should realize that “a” is the slope of the line.
- What is the derived unit associated with the value of “a”? Students should realize that neutrons are assigned to the y-axis, while protons are assigned to the x-axis. Thus, the derived unit is neutrons/proton.
- Examine your list. If you continue your list, what would be the next element? Student answers will vary depending on their list.
- Using the a-value that you obtained, predict the number of neutrons that the next element ought to have. Student answers will vary depending on their list.
- If you were to add the number of protons and neutrons together, do you obtain the mass number listed on the periodic table for that element? All students should realize that they get a mass number that is close to the one listed on the periodic table.
Drawing Conclusions: The following questions are to be answered as a class after each group has appropriately labeled their points on the class graph and labeled their slopes.
- As the atomic number increases, what happens to the value of “a”? The value of a gradually increases or become steeper.
- As the atomic number increases, what happens to the number of neutrons/proton? The number of neutrons required for every proton gradually increases.
- Why would atoms with higher atomic numbers need more neutrons? To stabilize the nucleus.
- What type of elements have the highest ratio of neutrons per proton? Students should tap into their prior knowledge and know determine that many elements with neutron/proton ratios of 1.5 or higher are radioactive elements.
- If a person has too much body weight and their doctor informs them that their weight is affecting the stability of their health, what is that person likely to do? Students should eventually suggest that a person would change their diet or exercise in order to lose weight.
- If an atom has too many protons and neutrons, what do you think the atom is likely to do? Based on the previous question, students should be able to determine that the atom will decide to get rid of protons and/or neutrons. Students may also suggest that atoms can become unstable just like a person’s health. Introduce to students that the forthcoming lessons will investigate nuclear decay processes.
Lesson(s): Alpha and Beta Decay
Purpose: The purpose of this lesson is to build upon the knowledge of the students’ use of nuclear notation and calculating subatomic particles by introducing alpha and beta decay. Students now know that the alpha process cannot create every element on the periodic table and they are left with a clue that some elements are created from a decay process based on the previous lesson. This lesson will look at why radioactive decay is important.
The lesson on alpha and beta decay can proceed similarly to the lesson on alpha process. Below are some classic examples of beta decay. It is suggested that alpha decay and beta decay should be split into two class periods so that students can experience success in one process before moving to the next.

Video and Short Discussion: This video and discussion can be used to reignite interest in radioactive decay, specifically beta decay. Post the following prompt as the kick-off activity: “Think about someone you know that has cancer, diabetes, or Alzheimer’s disease. How do you think Chemistry is related to detecting those diseases?”. Inform the students to prepare to share their answers on the condition that they feel comfortable talking about the people they know.
Discussion Questions
- Inform students that you will show a video about isotopes. Show students the following discussion questions: How can isotopes be used? How does the isotope function in this video? What type of particle does the isotope emit?
- Show the short YouTube video entitled “How Does a PET Scan Work?[3]
- After the video, ask if they need a replay of the video. Then poll the class for answers to the discussion questions.
- Explain to the students that the isotope in FDG is fluorine-18. Ask the students to find fluorine on the periodic table and discuss products that contain fluorine. When the students mention toothpaste, ask the students which type of fluorine is used in toothpaste. Is it fluorine-18 or fluorine-19? Which type of fluorine is used in medical imaging?
- Refer to the isotope puzzle cube answers from the previous activities. Explain to the students that fluorine-18 becomes oxygen-18 via positron emission as mentioned in the video.
- Students still may not know what a positron is. Ask them which subatomic particle does positron most likely remind them of? Students should suggest an electron. Thus, students should be able to gather that electrons can be forced to assume a positive charge and become a positron.
Formative Assessments
Build an Atom PhET Lab: This computer simulation is now available in HTML5 meaning that a computer no longer needs to download software like Java to use it. It is a great tool for students to learn atomic structure and to visualize atomic stability before teaching the concept. The simulation also includes an option that allows for nuclear notation and the net charge of the atom for use with this unit or with future units that involve ion formation.[4]
Modeling Radioactive Decay: Modeling is a great way to reinforce learning for students in the chemistry class. The Environmental Protection Agency has great resources that review the different types of radioactive decay using physical modeling.[5] Activities Noteworthy
Summative Assessments
Unit Tests and Quizzes: Students will be quizzed weekly, while tests are administered at the end of a unit. Both assessment methods primarily meet criterion (a), knowing and understanding, in which students must be able to explain scientific knowledge, apply scientific knowledge and understanding to solve problems set in familiar and unfamiliar situations, and analyze and evaluate information to make scientifically supported judgments.
Predicting Products and Pathways in Supernovae Explosions
This assessment will primarily focus on criterion B – inquiring and designing. Students will be given two scenarios. In the first scenario, students will be given a specific sequence of nuclear processes in order to determine what isotope is formed at the end of the list of processes. In the second scenario, students will be given the starting nuclide and the ending nuclide of a nuclide formed during a nuclear explosion. Students will be required to propose a mechanism. They will hypothesize the number of occurrences of each process and provide reasons why those processes should be involved in the formation of the second nuclide. They will be required to explain how to manipulate the variables and record the data. Finally, they will journal and summarize the steps involved in the figuring out the mechanism they proposed.
Constructed Response Topics
Criterion (d) Reflecting on the Impacts of Science: Allow students to choose an article and/or theme for a constructed response summary.
- Theme: Isotopes and their importance
- Theme: Nucleosynthesis – the impact of creating new nuclides
- Theme: Nuclear Medicine – the impact of nuclides in medicine
- Article: Designer Isotopes Push the Frontier of Science.[6]
- Article: Researcher Nabs Doubly Magic Tin Isotope, a North American First.[7]
- Article: Physicists Peer Closely at Radioactive Decay of a Rare Isotope at the Edge of Nuclear Existence.[8]
Students will write a summary of the article, emphasizing the impact that the discoveries described in the chosen article has on society. Since scientific writing can drastically differ from what students learn in their English classes with the traditional five-paragraph essays, the format of the writing so be simplified to two or three paragraphs. The first paragraph should introduce the discovery or problem and its applications. The second paragraph should focus on the benefits and disadvantages or strengths and weakness of the information presented in the article. This summary can also double as a formative assessment so that students can learn how to discuss and evaluate the implications of science in regards to solving specific problems and issues. Students will be assessed in the same strands of criterion (d) in the next assessment listed.
In My Element Project
Purpose: The purpose of this project is assess what has already been learned in this unit while creating a bridge to the next unit on electrons. Students will take on the identity of an atom and will have to convince the class that they are the most important element in the universe. They will be required to use old and new scientific language in order to explain, discuss, and evaluate the impacts that their element has on society.
Part I – Element Research Project: The element research ought to be conducted in class so that students have time to gather information and ask questions about the project as they are conducting research. Access to computers or internet technology (via library computer, school laptops, tablets, etc.) are necessary to make the most of this opportunity. Allow students 25 minutes of class time to research 3 elements that they are interested in doing. Do not allow any students to sign up to select an element until everyone has had 25 minutes of research time. This will allow students to have a backup element just in case another classmate has selected the same element prior to them. A paper sign-up sheet works best. Students should not be allowed to change elements because they should have completed enough research in 25 min to choose wisely.
The format for the research project requires a physical derivative work. The suggested formats are poem, song/video, brochure/pamphlet, or a one-page graphical information sheet. Regardless of the format, there must be a written or transcribed format. For example, if you write a poem, there must be a transcription of the poem with pictures. If the format is a video, then the video either needs to be accompanied with a script or the transcription can be included in the video itself. The video option is highly encouraged for students who may be uncomfortable with public speaking or with English language learners. Since Part I of this assessment is based on primarily facts and information, it will be assessed using strands from criterion (a), knowing and understanding.
Part I can also be assessed using criterion (d), reflecting on the impacts of science. Students will have to personify their element and convince classmates why they are the most important element that mankind will ever need. Students must present both the pros and the cons of their element and cite sources to support their argument.
Part II – Build an Atomic Model: The second task is for students to build a 3-dimensional Bohr model of the atom. Although the Bohr model is outdated, it still reinforces the concept that protons are neutrons reside in the nucleus while the electrons are located on various energy levels. Modern atomic theory states that the electron regions have complex shapes; therefore, it is not realistic to build an atomic model established on modern atomic theory. The construction of the atomic model focuses on the organization and presentation of information in a visual aid format. Thus, this portion of the project will be assessed using strands from criterion (c), processing and evaluating.
Part III – Presentations: Students will be allotted 5 minutes each to present their arguments to the class concerning the importance of their element to mankind. Classmates will evaluate their classmates on whether or not their arguments were convincing and will be required to cite evidence from the presentations to support their rating. The peer rating is necessary for managing classroom behaviors and to further enhance student learning. Essentially, the evaluation doubles as a personal set of notes with facts about each element presented. Students should not be expected to sit quietly for 25-30 presentations and become passive participants in this part of the project. This normally takes 2-3 days of classroom instruction.
Preplanning
Prior to the introduction of this project, students will have been introduced to the discovery of atoms and their subatomic particles. After the introduction to protons, neutrons, and electrons, students will receive the project rubric on a Monday. Students are given two days to overlook the rubric before selecting an element. On Wednesday, students will be provided with computer access to research their elements of interest for 30 minutes to determine if they can collect enough information to fulfill all the requirements of the rubric. After 30 minutes, students will be allowed to select their element on a first-come first-serve basis. Students will not be allowed to change elements unless absolutely necessary. Students are then given a week and a half to develop their project.
During the week before the atomic model is due, students will engage in an in-class activity to sketch and build two-dimensional models of elements that will further enhance their understanding of the role of the subatomic particles. Thus, before the final atomic model is due, students will have been assessed on the Bohr model of the atom by quiz and a classroom activity.
Addressing Diverse Learning Needs
The learning needs of students are each addressed in the 3 parts of the assessment. In Part I, students must produce a writing sample in the form of a poem, song, brochure, or an information sheet. This allows for students to think creatively on how to present the information collected. Students are given the option organize their thoughts in a non-traditional, written format that can make the project more fun. Students have opportunities to personify their elements using poems and songs. In addition, students who are more comfortable with traditional presentations have the option of creating an information sheet or poster. In Part II of the assessment, visual learners can become engaged through the craftsmanship of an atomic model to demonstrate their perception of what atoms look like on the microscopic level given that no human being actually knows what an atom actually looks like. Finally, in Part III, some students are best at communicating information orally rather than through artwork or written expression. Thus, there is plenty of opportunity for students to demonstrate their understanding of atoms outside of the traditional paper and pencil test.
In addition, research has shown that developing performance-based assessments with rubrics are a way of improving equity among students, specifically with English-language learners. Siegel argues that rubrics clearly inform students what is expected of them as well as they cause the teacher to focus on the equity in its design.[9] The rubric can actually serve as a feedback tool for teachers consider modifications of the rubric, which further promotes equity. The rubric also sets up the scaffold for using scientific language to support student learning. Since this assessment is heavily focused on the subatomic particles and their functional roles, this project shows all students the context of the language that is being required, regardless if the student is an ELL or a general education student.
Post-Project Learning
Upon successful completion of the project, students will have a gained an introduction to the periodic table of elements that will allow them to relate to the elements on a personal as well as a basic level before engaging into more complex structure/function relationships between the three types of subatomic particles. Prior to the conclusion of this unit, students will have acquired understanding of 60% of the requirements demanded in the rubric, giving the students opportunity to visually demonstrate the basics of protons, neutrons, and electrons. Their research will extend their introductory knowledge of electrons, such as the number of electrons in each shell. Thus, students’ completion of their project could propel them into higher levels of engagement and seeking understanding of the elements on the periodic table in the future. Also, since students are responsible for the presentation of their individual elements, this will promote healthy student-centered learning as the students are given the opportunity to educate peers while the teacher simply facilitates. Finally, given that this performance-based assessment marks the beginning of which students will begin to dive into the deeper aspects of chemistry, this assessment helps students to identify forthcoming vocabulary and scientific terms that need to be translated and retained throughout the remainder of the course.
The project deadlines may be adjusted according to your curriculum needs. For example, the project deadline can occur at the conclusion of this unit, in the midst of the second atomic structure unit focusing on electrons, or at the end of the second unit after electrons have been thoroughly covered.
Isotopes Formed in Supernovae Explosions
Students will be provided with a list of isotopes formed in supernovae explosions and their decay products. Students will be required to identify the processes and the frequency of their occurrences to determine nuclear pathway by which the SNE isotope becomes a stable product. The following table includes a lists of nuclides that could be used in the assessment. This activity can be assessed using criterion (a) and criterion (b).
| List of Isotopes and their Products from Supernovae Explosions[10] |
| SNE Isotope |
Product Isotope |
Decay Particles |
| 41Ca |
41K |
Electron capture |
| 60Fe |
60Ni |
2 beta emissions |
| 26Al |
26Mg |
1 positron emission |
| 237Np |
209Bi |
7 alpha emissions, 4 beta emissions |
| 53Mn |
53Cr |
Electron capture |
| 129I |
129Xe |
1 beta emission |
| 236U |
232Th |
1 alpha emission |
| 235U |
207Pb |
7 alpha emissions, 4 beta emissions |
| 238U |
206Pb |
8 alpha emissions, 6 beta emissions |
[1] Ventuno Art. (2015). How to Fold an DIY : Origami 3D Cube. Retrieved August 18, 2016, from https://www.youtube.com/watch?v=337QxhfpY4w
[2] ACS Reactions. (2015). The Science of the Avengers. Retrieved August 17, 2016, from https://www.youtube.com/watch?v=Gr3ov7R89Xo
[3] NIBIB gov. (2013). How Does a PET Scan Work? Retrieved August 17, 2016, from https://www.youtube.com/watch?v=GHLBcCv4rqk
[4] Build an Atom. (n.d.). Retrieved August 17, 2016, from https://phet.colorado.edu/en/simulation/build-an-atom
[5] Radiation Education Activities. (n.d.). Retrieved August 17, 2016, from https://www3.epa.gov/radtown/educational-materials.html
[6] Michigan State University. (2008, May 9). Designer Isotopes Push The Frontier Of Science. ScienceDaily. Retrieved August 17, 2016 from www.sciencedaily.com/releases/2008/05/080508164631.htm
[7] National Superconducting Cyclotron Laboratory at Michigan State University. “Researcher Nabs Doubly Magic Tin Isotope, A North American First.” ScienceDaily. ScienceDaily, 24 December 2008. www.sciencedaily.com/releases/2008/12/081210145307.htm
[8] Michigan State University. “Physicists Peer Closely At Radioactive Decay Of A Rare Isotope At The Edge Of Nuclear Existence.” ScienceDaily. ScienceDaily, 15 November 2007. www.sciencedaily.com/releases/2007/11/071108155357.htm
[9] Siegel, M. (2014). Developing Preservice Teachers’ Expertise in Equitable Assessment for English Learners. Journal of Science Teacher Education, 25(3), 289-308. doi: 10.1007/s10972-013-9365-9
[10] Possibility of life origin on radioactive source of energy after our supernova explosion. (n.d.). August 17, 2016, http://dx.doi.org/10.1117/12.375080

![]() Each group of 6 should try to solve for each missing component of the nuclear symbol. Once each group finishes the 6 nuclear symbols, they should trade cubes with another group. Have the groups continue trading until every group has completed the symbols of every other group.
Each group of 6 should try to solve for each missing component of the nuclear symbol. Once each group finishes the 6 nuclear symbols, they should trade cubes with another group. Have the groups continue trading until every group has completed the symbols of every other group.![]() .
.![]() . Ask the students what could have happened to the 2 protons. Students ought to respond saying that two protons must have become neutrons. Afterward, introduce the students to gamma emission, which is the energy released as photon when a proton transforms into a neutron. Explain to students that this process is the beginning of stellar nucleation (star creation) and that the first step of stellar nucleation produces the alpha particle, which is key in the alpha process. Model for the students how to write a nuclear reaction equation using the four protons.
. Ask the students what could have happened to the 2 protons. Students ought to respond saying that two protons must have become neutrons. Afterward, introduce the students to gamma emission, which is the energy released as photon when a proton transforms into a neutron. Explain to students that this process is the beginning of stellar nucleation (star creation) and that the first step of stellar nucleation produces the alpha particle, which is key in the alpha process. Model for the students how to write a nuclear reaction equation using the four protons.![]() . The equation is as follows:
. The equation is as follows: ![]() . Then ask to students to write the nuclear chemical reaction of the nuclides involved in the diagram. Have the students report out their answer and explain why they believe the nuclides they have written are what matches in the diagrams. Challenge the students to write a rule or journal the steps involved in writing the nuclear notation for nuclides produced in the alpha process. Students should have a procedure similar to the following steps:
. Then ask to students to write the nuclear chemical reaction of the nuclides involved in the diagram. Have the students report out their answer and explain why they believe the nuclides they have written are what matches in the diagrams. Challenge the students to write a rule or journal the steps involved in writing the nuclear notation for nuclides produced in the alpha process. Students should have a procedure similar to the following steps: